Introduction
To begin the year, our teacher taught us how elements were first created because they will be the building blocks of everything we will be learning this year. This basically meant we started at what humans believe to be the beginning of the universe, the Big Bang, because this is went the first atoms were created. The very first thing we did was make a class model of the timeline of the early stages of the universe after the Big Bang. After establishing a basic idea of the order, we learned more about the details such as how the periodic table grew over time, the life cycle of stars, structures of atoms, and decay. In order to show what we learned, we created various models and took notes on each subject. At the end, each group chose a specific subject to focus on, and create some sort of unique model to represent that subject.
Terms
atom
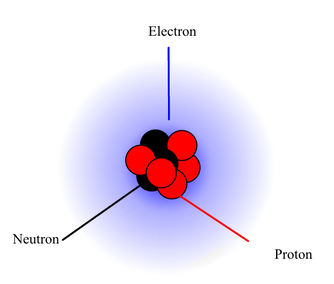
This is the smallest particle an element can exist in, however it consists of smaller particles, protons, neutrons, and electrons.
Size: 10^-10m
Size: 10^-10m
|
Vertical Divider
|
<
>
A particle in an atom that is located in the nucleus. The number of these particles determine the element the atom is and its atomic number on the periodic table. When the number of protons is added to the number of neutrons, it results in the mass of that atom. An element will always have the same number of protons in it. Charge: Positive Mass: 1.oo728 amu Size: 10⁻¹⁵ m Symbol: P⁺ This is a particle in the atom that is located in the nucleous. When the number of neutrons is added to the number of protons, it results in the mass of the atom. The number of neutrons in an element is not always consistent. Charge: Neutral Mass: 1.00866 amu Size: 10⁻¹⁵ m Symbol: n⁰ This is the core of the atom where the protons and the neutrons reside. It is also the densest part of the atom and is where all the mass of the element comes from. However, this makes up very little of the atom's volume. The mass of the nucleus is not always consistent with the element. It is a somewhat particle and also not particle of an atom that has not quite been confirmed of its actual form. The mass of these are so small that they are not included when calculating the mass of the atom. However, they extend so far that they pretty much make of the entire volume of the atom. The number electrons is not consistent with elements. Charge: Negative Mass: 0.000055 amu Size: 10⁻¹⁸ Symbol: e⁻ |
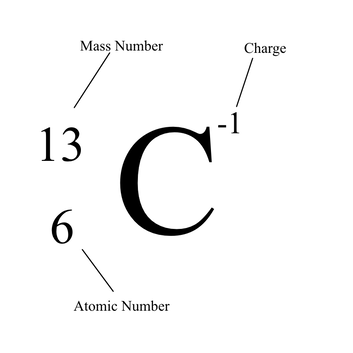
Isotopic Notation
|
This is a way of writing out the basic information about an atom in a simplified way for efficiency.
<
>
This tells us the mass of the atom that is being talked about at the moment. The number is the number of protons added to the number of neutrons,. It will always be whole because there cannot be part of a proton or a neutron. This is not always the same for atoms of the same element. This is the number the element is on the periodic table. It is also the number of protons an element has. The number here will never change for atoms of the same element. This is the symbol used to represent the element. It will either be one upper case letter or one upper case letter and a lower case letter. Typically these are based off the name of the element but not always. This never changes for atoms of the same element, and no two elements will have the same symbol. This tells us the charge of the atom being shown. If there is no number there, it has a balanced or neutral charge. When there is a negative sign followed by a number, it has a negative charge. However, if there is a positive sign followed by a number, that means that the atom has a positive charge. This calculated by subtracted the number of electrons by the number of protons. This is not the same for each atom of the same element. |
Vertical Divider
|
Isotope
Atoms of an element that have the same number of protons but a different number of neutrons. This results in the atoms having different masses. The average masses of these isotopes is represented as the mass of the elements on the periodic table.
Ion
When atoms of the same element have the same number of protons, but a different number of electrons. This results in the charge being different. If the number of protons and electrons are the same, then the atom is neutral because it has no charge.
Radioactive decay
The spontaneous breakdown of an atom's nucleus that results in a release of energy and matter from the nucleus. There are three types, alpha, beta, and gamma.
Alpha Decay
A types of radioactive decay that results in the release of an alpha particle. An alpha particle is a helium atom that has a mass of 4, an atomic number of 2, and no charge.
Beta Decay
The type of radioactive decay that results in the release of a beta particle. A beta particle is shown as either an e or a beta symbol with a mass of zero, one negative proton, and no charge.
Gamma Decay
The type of radioactive decay that results in the release of a y-ray, which is high-energy radiation that releases a particle that is represented by a Y with no mass, no protons, and no charge.
Positron Emission
A type of decay that results in emitting a positron. This is represented by an e with a mass of zero, one proton, and no charge.
NUCLEAR TRANSMutation
When an unstable atom changes its nucleus by emitting energy and mass which results in a different element.
NUCLEOSYNTHESIS
The creation of atoms that are not hydrogen.
FISSON
When two elements are created from the splitting of an atom and a release of energy,
FUSION
When a new atom is created from the collision of two atoms and their nucleus fusing,
Project
Description
For the model, my group decide to make a stop motion animation in the South Park style. The video goes over the three different types of radioactive decay, beta, alpha, and gamma. It first began with researching the types of decay in depth to decide which information was most important to put in the video. The second part was creating the script with all the lines. Both are shown below.
For the model, my group decide to make a stop motion animation in the South Park style. The video goes over the three different types of radioactive decay, beta, alpha, and gamma. It first began with researching the types of decay in depth to decide which information was most important to put in the video. The second part was creating the script with all the lines. Both are shown below.
Background
|
Research
|
Script
|
Video
Reflection
Overall the project was adequate. A few things that went well was the initial research and script writing. However, we could have improved on our timing more and splitting up the jobs more for the final part of the project. For this project we were really able to show our collaboration and creativity. Two things we could work on though is communication and consciousness learning.
When researching the different types of decay, it went well because each person had something to research and were able to take notes on their research in a way their group members could understand. Dividing up the work here worked because it made sure we did not research the same thing four times and allowed the work to be completed faster. Since some of our group members had an idea of what they wanted the video to look like they wrote the outline of the script, while the people who did research, put their information in according to where it was on the script. This is a better example to show how dividing the work helped us because it was the combining of two pieces that were previously worked on separately. Another positive was that it allowed each team member to have a job to do while still contributing to the end product.
Time management could have been a lot better for this project because at one point we had all the pieces, but didn't find the time to put them all together. So the project itself was late because of extensions and things such as the blackout, when we probably could have gotten more done earlier if we managed our time a bit better. Unlike the first half of our project, the second half really relied on part of the team because the other team members didn't quite grasp the vision of the video. When it really should have been everyone working on the project, I really should have been there more to help out with whatever they needed help with to finish the project on time. If I could change what we did, I would probably get the recording done earlier and done more stop motion filming earlier to get things done. Something else is probably sending people out to record voice overs while the other team members are filming the stop motion just to make sure everyone is doing something during class.
For the first half at least, the team was very collaborative with each other and doing what they could to add on to the project. An example of this is the research, each person had something they were suppose to be working on so that at the end we all had the correct information. This allowed work to be done quicker and more efficiently. This model was very creative seeing as how it has Jesus and MC Dub even though it is a video about radioactive decay. It was very ambitious with what we were trying to go for and pretty much succeed in explaining what we needed to explain. Communication could have been worked on a bit because everyone was pretty much quiet when we probably could have talked more about when we would be working on and finishing the project. Had we talked out timing a bit more we might have finished it more on time than we did. My consciousness learning can be improved because I should have volunteered more to help out with completing the video even though I don't know anything about editing videos, I could have learned. At some points I could tell I wasn't doing much for the video and should have pushed my learning farther and asked how I could have helped with completing it.
When researching the different types of decay, it went well because each person had something to research and were able to take notes on their research in a way their group members could understand. Dividing up the work here worked because it made sure we did not research the same thing four times and allowed the work to be completed faster. Since some of our group members had an idea of what they wanted the video to look like they wrote the outline of the script, while the people who did research, put their information in according to where it was on the script. This is a better example to show how dividing the work helped us because it was the combining of two pieces that were previously worked on separately. Another positive was that it allowed each team member to have a job to do while still contributing to the end product.
Time management could have been a lot better for this project because at one point we had all the pieces, but didn't find the time to put them all together. So the project itself was late because of extensions and things such as the blackout, when we probably could have gotten more done earlier if we managed our time a bit better. Unlike the first half of our project, the second half really relied on part of the team because the other team members didn't quite grasp the vision of the video. When it really should have been everyone working on the project, I really should have been there more to help out with whatever they needed help with to finish the project on time. If I could change what we did, I would probably get the recording done earlier and done more stop motion filming earlier to get things done. Something else is probably sending people out to record voice overs while the other team members are filming the stop motion just to make sure everyone is doing something during class.
For the first half at least, the team was very collaborative with each other and doing what they could to add on to the project. An example of this is the research, each person had something they were suppose to be working on so that at the end we all had the correct information. This allowed work to be done quicker and more efficiently. This model was very creative seeing as how it has Jesus and MC Dub even though it is a video about radioactive decay. It was very ambitious with what we were trying to go for and pretty much succeed in explaining what we needed to explain. Communication could have been worked on a bit because everyone was pretty much quiet when we probably could have talked more about when we would be working on and finishing the project. Had we talked out timing a bit more we might have finished it more on time than we did. My consciousness learning can be improved because I should have volunteered more to help out with completing the video even though I don't know anything about editing videos, I could have learned. At some points I could tell I wasn't doing much for the video and should have pushed my learning farther and asked how I could have helped with completing it.